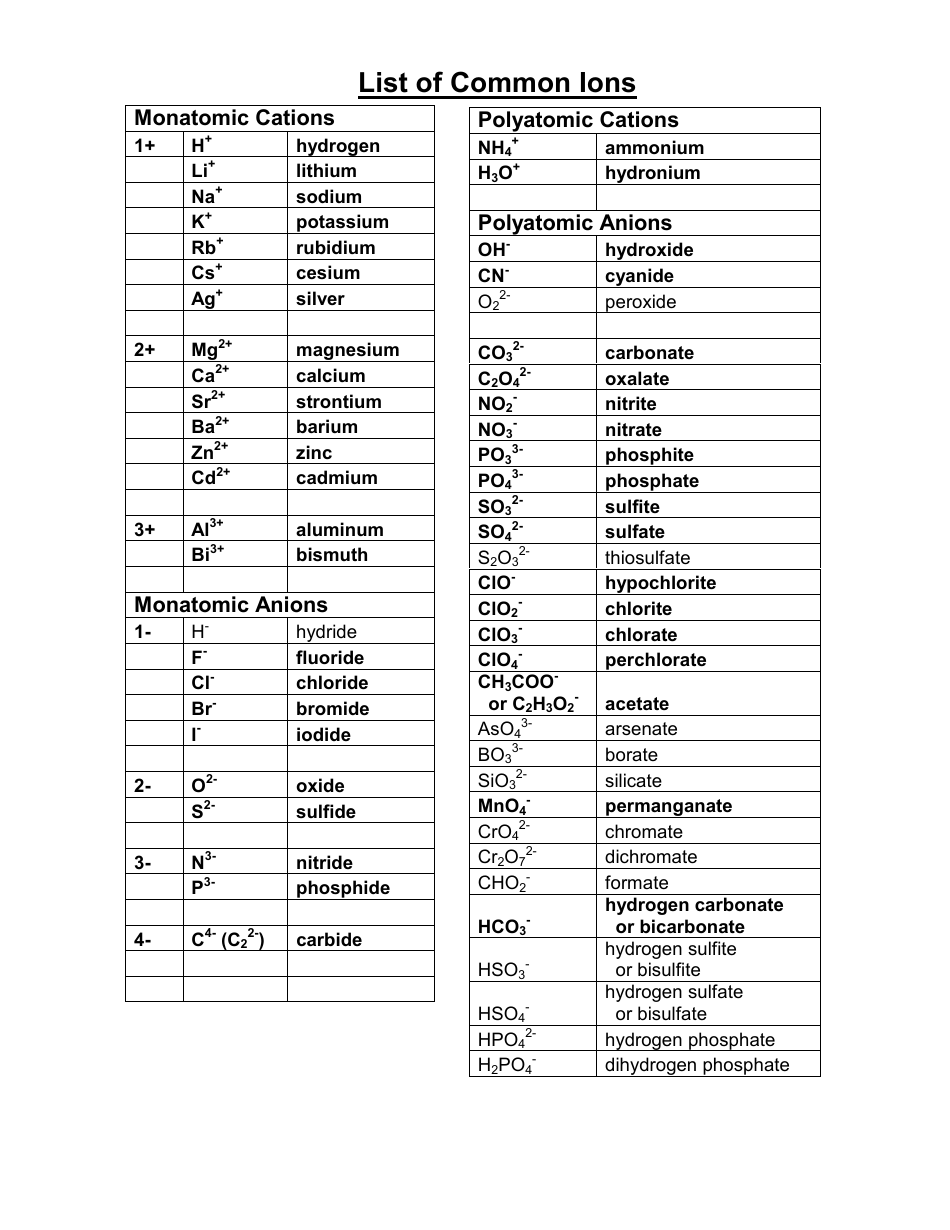
Anions Chart
Anions Chart - Ions consist of either atoms or groups of atoms that have a charge that is either negative or positive. They gain one or more than one electron. Anions are formed when atoms or molecules gain electrons to achieve a. Ions are charged atoms or molecules. They accept one or more electrons and. See examples of the two types of ions, learn how they are different, and see periodic table trends. Cations are ions that are positively charged. Anions are negatively charged ions that form when atoms gain electrons, and a list of anions includes common ones like chloride, sulfate, and nitrate. This table lists common anions by their names, with the anion formula and charge. Anions are so named because they are attracted to the anode (positive field) in an electrical field. Anions are negatively charged ions. In many crystals the anions are bigger; They accept one or more electrons and. This table lists common anions by their names, with the anion formula and charge. Ions are charged atoms or molecules. Anions are ions that are negatively charged. Anions are formed when atoms or molecules gain electrons to achieve a. Cations are ions that are positively charged. Therefore, they possess a net negative charge. The little cations fit into the spaces between them. This table lists common anions by their names, with the anion formula and charge. The little cations fit into the spaces between them. See examples of the two types of ions, learn how they are different, and see periodic table trends. Anions are formed when atoms or molecules gain electrons to achieve a. Ions consist of either atoms or groups. Anions are termed so because they are attracted towards the anode (the positive electrode). In many crystals the anions are bigger; Anions are negative ions that are formed when a nonmetal atom gains one or more electrons. Anions are so named because they are attracted to the anode (positive field) in an electrical field. Ions consist of either atoms or. Cations are ions that are positively charged. See examples of the two types of ions, learn how they are different, and see periodic table trends. Anions are formed when atoms or molecules gain electrons to achieve a. Ions consist of either atoms or groups of atoms that have a charge that is either negative or positive. Therefore, they possess a. Anions are negatively charged ions that form when atoms gain electrons, and a list of anions includes common ones like chloride, sulfate, and nitrate. Anions are formed when atoms or molecules gain electrons to achieve a. In many crystals the anions are bigger; Get the definitions of cations and anions. Anions are so named because they are attracted to the. See examples of the two types of ions, learn how they are different, and see periodic table trends. Anions are negatively charged ions that form when atoms gain electrons, and a list of anions includes common ones like chloride, sulfate, and nitrate. Anions are ions that are negatively charged. Anions are negative ions that are formed when a nonmetal atom. Cations are ions that are positively charged. An anion is a negatively charged ion, meaning it has gained one or more electrons during a chemical reaction. Anions are negatively charged ions. Anions are negative ions that are formed when a nonmetal atom gains one or more electrons. Get the definitions of cations and anions. They accept one or more electrons and. Get the definitions of cations and anions. Anions are grouped according to whether they are simple (one element or monatomic ions),. Cations are ions that are positively charged. Anions are formed when atoms or molecules gain electrons to achieve a. In many crystals the anions are bigger; Anions are negative ions that are formed when a nonmetal atom gains one or more electrons. Get the definitions of cations and anions. Anions are formed when atoms or molecules gain electrons to achieve a. An anion is a negatively charged ion, meaning it has gained one or more electrons during a chemical. This table lists common anions by their names, with the anion formula and charge. Anions are negatively charged ions. In many crystals the anions are bigger; The little cations fit into the spaces between them. Cations are ions that are positively charged. Anions are negative ions that are formed when a nonmetal atom gains one or more electrons. Ions consist of either atoms or groups of atoms that have a charge that is either negative or positive. Anions are so named because they are attracted to the anode (positive field) in an electrical field. Therefore, they possess a net negative charge. Anions. This table lists common anions by their names, with the anion formula and charge. In many crystals the anions are bigger; They gain one or more than one electron. Anions are negative ions that are formed when a nonmetal atom gains one or more electrons. See examples of the two types of ions, learn how they are different, and see periodic table trends. An anion is a negatively charged ion, meaning it has gained one or more electrons during a chemical reaction. Anions are negatively charged ions that form when atoms gain electrons, and a list of anions includes common ones like chloride, sulfate, and nitrate. Anions are so named because they are attracted to the anode (positive field) in an electrical field. Anions are ions that are negatively charged. Cations are ions that are positively charged. Anions are grouped according to whether they are simple (one element or monatomic ions),. Anions are negatively charged ions. Get the definitions of cations and anions. Ions are charged atoms or molecules. Anions are formed when atoms or molecules gain electrons to achieve a. Anions are termed so because they are attracted towards the anode (the positive electrode).Science 9 Chapter 3 Ions, Polyatomic ions Examples of Some Cations and Anions with valency
Cations and Anions List KarsyntinOconnell
Cations And Ions Explained Chart
Common ions anions and cations Artofit
Table of Anions
Common Ions Chart Download Printable PDF Templateroller
Common Anions List and Formulas
Cations And Anions Chart A Visual Reference of Charts Chart Master
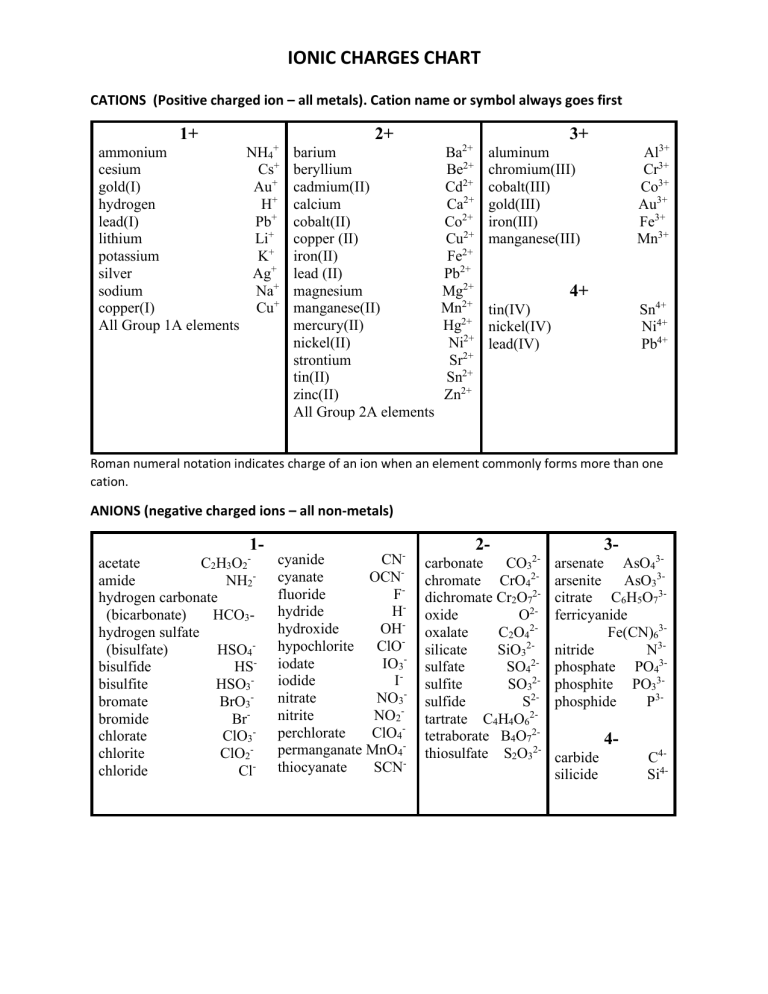
Ionic Charges Chart Cations & Anions
two tables showing the different types of common anions
Ions Consist Of Either Atoms Or Groups Of Atoms That Have A Charge That Is Either Negative Or Positive.
They Accept One Or More Electrons And.
Therefore, They Possess A Net Negative Charge.
If They Have Have Gained Electrons, They Will Be Negatively Charged, Due.
Related Post: