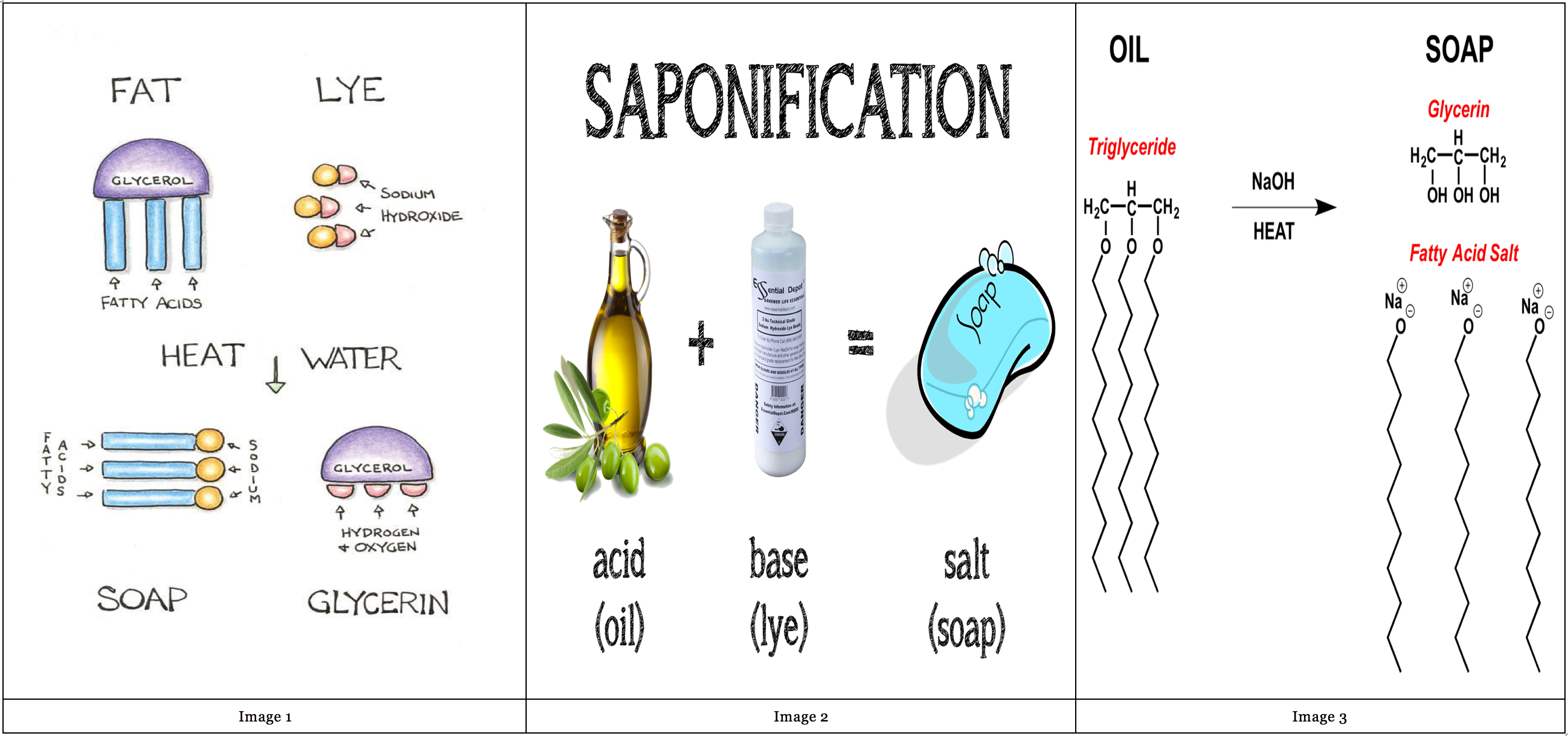
Saponification Chart
Saponification Chart - This process is called basic hydrolysis of esters. The alkali used in this process is sodium hydroxide. Typically aqueous sodium hydroxide solutions are used. Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called soap. Esters can be cleaved back into a carboxylic acid and an alcohol by reaction with water and a base. Saponification is the fundamental chemical reaction in soap making where triglycerides from fats or oils react with a strong alkali, typically sodium hydroxide or potassium. Saponification is a process of cleaving esters into carboxylate salts and alcohols by the action of aqueous alkali. Saponification is the hydrolysis of an ester to form an alcohol and the salt of a carboxylic acid in acidic or essential conditions. This process involves hydrolysis, where water molecules. Saponification is a chemical reaction between a strong base and a triglyceride that results in the formation of a salt. The alkali used in this process is sodium hydroxide. Saponification is the fundamental chemical reaction in soap making where triglycerides from fats or oils react with a strong alkali, typically sodium hydroxide or potassium. This process involves hydrolysis, where water molecules. In this article, we will explain the saponification. Saponification is the hydrolysis of an ester to form an alcohol and the salt of a carboxylic acid in acidic or essential conditions. Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called soap. Saponification is a chemical reaction between a strong base and a triglyceride that results in the formation of a salt. When esters are treated with hydroxide ion, followed by neutralization with acid, they are converted into carboxylic acids. Typically aqueous sodium hydroxide solutions are used. This process is called basic hydrolysis of esters. Typically aqueous sodium hydroxide solutions are used. Saponification is a chemical process in which triglycerides undergo an interaction with lye made from sodium or potassium hydroxide resulting in glycerol as well as a fatty acid. Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called soap.. Saponification is the fundamental chemical reaction in soap making where triglycerides from fats or oils react with a strong alkali, typically sodium hydroxide or potassium. Esters can be cleaved back into a carboxylic acid and an alcohol by reaction with water and a base. The reaction is called a saponification from the latin sapo which means soap. Saponification is a. Saponification is a process of cleaving esters into carboxylate salts and alcohols by the action of aqueous alkali. When esters are treated with hydroxide ion, followed by neutralization with acid, they are converted into carboxylic acids. Typically aqueous sodium hydroxide solutions are used. In this article, we will explain the saponification. The reaction is called a saponification from the latin. Saponification is a chemical process in which triglycerides undergo an interaction with lye made from sodium or potassium hydroxide resulting in glycerol as well as a fatty acid. Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called soap. Saponification is the hydrolysis of an ester. Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called soap. The alkali used in this process is sodium hydroxide. Esters can be cleaved back into a carboxylic acid and an alcohol by reaction with water and a base. Saponification is the hydrolysis of an ester. Saponification is a process of cleaving esters into carboxylate salts and alcohols by the action of aqueous alkali. Typically aqueous sodium hydroxide solutions are used. Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called soap. When esters are treated with hydroxide ion, followed by neutralization. Saponification is a chemical process in which triglycerides undergo an interaction with lye made from sodium or potassium hydroxide resulting in glycerol as well as a fatty acid. Saponification is the fundamental chemical reaction in soap making where triglycerides from fats or oils react with a strong alkali, typically sodium hydroxide or potassium. Saponification is a chemical reaction between a. When esters are treated with hydroxide ion, followed by neutralization with acid, they are converted into carboxylic acids. Typically aqueous sodium hydroxide solutions are used. Saponification is the hydrolysis of an ester to form an alcohol and the salt of a carboxylic acid in acidic or essential conditions. Saponification is a process of cleaving esters into carboxylate salts and alcohols. The alkali used in this process is sodium hydroxide. Saponification is the fundamental chemical reaction in soap making where triglycerides from fats or oils react with a strong alkali, typically sodium hydroxide or potassium. Typically aqueous sodium hydroxide solutions are used. When esters are treated with hydroxide ion, followed by neutralization with acid, they are converted into carboxylic acids. This. Saponification is a process of cleaving esters into carboxylate salts and alcohols by the action of aqueous alkali. In this article, we will explain the saponification. The alkali used in this process is sodium hydroxide. Saponification is a chemical reaction between a strong base and a triglyceride that results in the formation of a salt. When esters are treated with. Saponification is a process by which triglycerides are reacted with sodium or potassium hydroxide (lye) to produce glycerol and a fatty acid salt called soap. Saponification is a chemical reaction between a strong base and a triglyceride that results in the formation of a salt. Saponification is a process of cleaving esters into carboxylate salts and alcohols by the action of aqueous alkali. The reaction is called a saponification from the latin sapo which means soap. This process involves hydrolysis, where water molecules. In this article, we will explain the saponification. Esters can be cleaved back into a carboxylic acid and an alcohol by reaction with water and a base. This process is called basic hydrolysis of esters. Saponification is the hydrolysis of an ester to form an alcohol and the salt of a carboxylic acid in acidic or essential conditions. When esters are treated with hydroxide ion, followed by neutralization with acid, they are converted into carboxylic acids. Typically aqueous sodium hydroxide solutions are used. The alkali used in this process is sodium hydroxide.Soap saponification charts information natural health Artofit
Faire son savon soimême la saponification à froid Bioté Naturelle
Helping Vocabulary to Stick 7 Successful Strategies for Schooling Saponification (or at least
Soap Saponification Charts & Information Natural Health
Soap saponification charts information natural health Artofit
Soap Saponification Charts & Information Cold process soap recipes, Soap making recipes
أغسطس 2018 sciences
Free fatty acid content, Saponification Value and Kinematic viscosity... Download Table
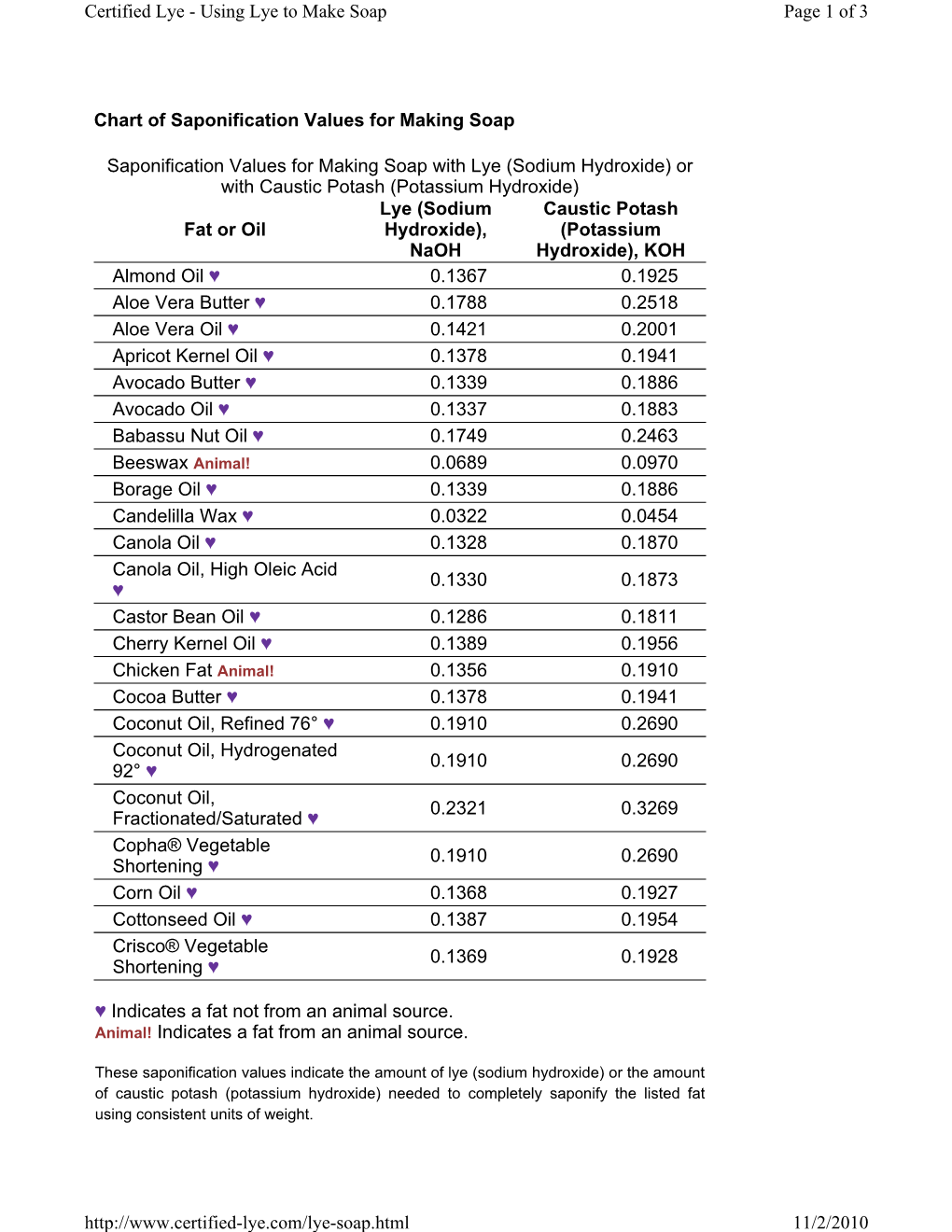
Chart of Saponification Values for Making Soap Fat Or Oil DocsLib
Average chain length and saponification values for common edible oils Download Scientific Diagram
Saponification Is A Chemical Process In Which Triglycerides Undergo An Interaction With Lye Made From Sodium Or Potassium Hydroxide Resulting In Glycerol As Well As A Fatty Acid.
Saponification Is The Reaction Where An Ester Is Hydrolyzed Into An Alcohol And A Carboxylic Acid Salt Upon The Addition Of An Aqueous Base.
Saponification Is The Fundamental Chemical Reaction In Soap Making Where Triglycerides From Fats Or Oils React With A Strong Alkali, Typically Sodium Hydroxide Or Potassium.
Related Post: